Cancer is a significant global health problem characterized by the uncontrolled proliferation, growth, and invasion of cancer cells beyond their usual boundaries into nearby tissues. It substantially threatens public health worldwide, leading to increased mortality and disability. In 2020, the World Health Organization (WHO) estimated that there were over 19.3 million new cancer cases and 10 million cancer-related deaths. Alarmingly, it is projected that by 2030, approximately 30 million people will succumb to cancer annually. All this points to the fact that the potential of molecular diagnostics for cancer research must be explored.
Given the multifaceted nature of cancer, finding suitable treatment options remains an ongoing challenge. Therefore, there is an urgent need to focus on early detection and appropriate treatment to improve patient outcomes and enable healthcare professionals to make well-informed decisions regarding patient care.
Molecular diagnostics has emerged as a widely accepted and reliable platform within the field of in vitro diagnostics. It offers the highest level of diagnostic accuracy and plays a crucial role in assessing the risk of developing cancer. By analyzing genetic markers and alterations, molecular diagnostics can provide valuable insights into an individual’s predisposition to cancer. Furthermore, this approach has the potential to differentiate between cancerous and benign tumors and even determine the tissue type from which the cancer originated.
Molecular diagnostics in the early detection of cancer
The success of cancer treatment greatly relies on early detection. Prevention and early detection are about combating cancer before the disease becomes too difficult to treat. In the UK, patients diagnosed with lung cancer have an 83% chance of one-year survival if the cancer is detected at stage 1. However, this drops significantly to just 17% if the cancer is diagnosed in the latter stage. Thus, researchers have turned their attention to developing low-cost, high-throughput, early-detection tools with high selectivity and specificity for cancer detection.
Tools and techniques for early cancer detection
Molecular analysis of the oncogenes and tumor suppressor genes involved in particular types of tumors can provide significant information for cancer diagnosis and in monitoring the effects of treatment. In recent times, molecular diagnosis is emerging as a new and eye-opening approach that merges genomics and proteomics for early detection and diagnosis of cancer.
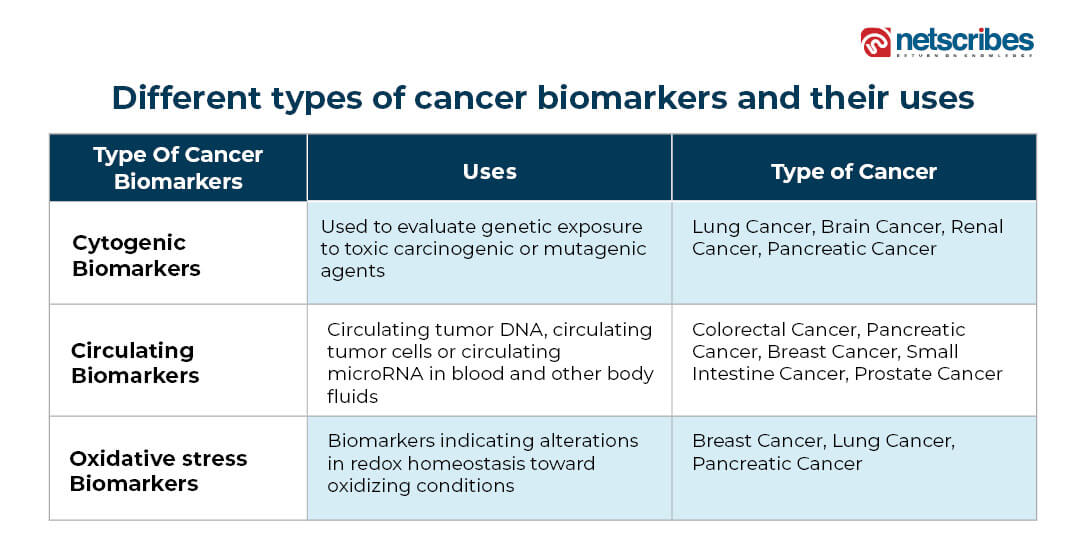
Biomarkers for early cancer detection
Molecular biomarkers are alterations in chromosomes, DNA, RNA, proteins, and related molecules that indicate a specific biological condition, such as cancer. These biomarkers have various applications, including assessing an individual’s risk of developing cancer, predicting its recurrence, and monitoring disease progression to evaluate the effectiveness of therapy.
As tumors progress, molecular changes occur, and genomic, proteomic, and metabolomic biomarkers can all potentially be utilized to detect cancer, determine prognosis, and monitor disease progression and response to treatment.
Liquid biopsy
It is a non-invasive approach that allows real-time monitoring of cancerous progression. Liquid biopsies use blood samples to identify specific biologic signals in DNA, RNA, or proteins released by cancer cells into the blood. It is a Next Generation Sequencing (NGS)-based test that employs AI algorithms to analyze methylation patterns of cell-free DNA (cfDNA) in blood. Liquid biopsies can detect:
Circulating tumor DNA: Circulating tumor is an early detection biomarker for cancers since tumors secrete ctDNA into the bloodstream before they are visible on imaging, signs of disease are detected, or both. Moreover, ctDNA testing may be synergistically used with other multi-omic biomarkers to enhance early detection. For instance, assays may incorporate early detection proteins (i.e., CA-125), epigenetic markers, circulating tumor RNA, nucleosomes, exosomes, and associated immune markers. ctDNA levels are higher in patients at advanced stages of breast, colorectal, pancreatic, and gastro-esophageal cancer than in early-stage patients.
Circulating tumor cells (CTCs): Circulating tumor cells (CTCs) are shed into the bloodstream from primary tumors during the early stages of tumorigenesis. A major application of CTCs is early tumor detection and prediction of metastasis. A fluid-phase biopsy reveals CTC prevalence rates and morphologic heterogeneity in early-stage NSCLC.
In addition to this, Real-time PCR, digital PCR, MLPA, fragment analysis, sanger sequencing, pyrosequencing, and next generation sequencing(NGS) are some of the widely accepted methods in early cancer detection.
Integration of personalized medicine with molecular diagnostics tests in clinical practice
In the past decade, several druggable tumor-specific molecular aberrations have grown substantially due to significant survival benefit obtained from biomarker-matching therapies in numerous cancer types. Therefore, molecular pathology has become essential not only for tumor diagnosis but also to drive therapeutic decisions in daily practice. The rising number of large-scale tumor profiling programs across institutions along with the introduction of next-generation sequencing technologies have revolutionized the field of precision oncology.
Precision medicine tools like immunohistochemistry (IHC) and fluorescence in situ hybridization (FISH) are widely used to detect biomarkers.
IHC can detect changes at the protein level that result from DNA rearrangements or point mutations and gene aberrations, the use of IHC determines the eligibility of therapies based on anti-PD-1/PD-L1 agents in certain tumor types.
FISH has been the gold-standard technique for DNA rearrangements, and it is routinely used to confirm gene level amplification when IHC results are ambiguous.
With the substantial growth of predictive biomarkers and druggable gene aberrations in oncology, conventional techniques have increasingly been substituted by NSG technologies as it provides more efficient, cost-and-tissue-saving tumor analysis as compared to single biomarker analysis.
Due to the decreasing cost and turnaround time of NGS, synchronization of knowledgebases for the clinical interpretation of genomic results, and the shift towards NGS in clinical testing is instrumental in the precision cancer medicine context.
Related reading: Navigating the future of infectious disease control with molecular diagnostics
Role of molecular diagnostics in monitoring treatment response
Monitoring response to treatment is a key element in the management of cancer. Molecular diagnostics, which can be repeated multiple times over the course of treatment such as simple blood tests, have been developed for colon and several other cancers to examine treatment response.
Some cancers might develop resistance to medications, as in the case of chronic myelogenous leukemia which is treated with a medication known as imatinib. Generally, patients take imatinib for years, however, in some cases the sequence of genetic material can change over time, which can further lead to reduced effectiveness of the medication. Consequently, such patients may undergo molecular diagnostic testing to determine whether the gene has changed.
Sequential monitoring of ctDNA plays a key role in assessing the treatment response or monitoring relapse. Genomic studies reveal that analysis of ctDNA for driver mutations in e.g. EGFR, KRAS, and BRAF could be used for treatment selection in addition, it has the potential to monitor treatment response, detect early residual disease, and identify drug resistance mechanisms. ctDNA provides information about disease status on a real-time basis, as it has a short half-time (~1-2 hours), also it possesses tumor-specific attributes. On the contrary, protein markers are not tumor-specific and have longer half-life times, which can further limit their added value in monitoring treatment response.
Advancements and future directions in molecular diagnostics in cancer research & care
Over the last few decades, advancement in molecular diagnostics has contributed to significant progress in cancer research and diagnostics, by dramatically improving lab workflows.
Advanced platforms including next-generation sequencing(NGS), and immunotherapies like checkpoint inhibitors are now a reality for cancer patients. NGS has emerged enormously for drug development and discovery and can also be used to understand disease biology. Moreover, NGS is also helpful in discovering mechanisms of drug resistance and rational drug combinations.
Recently, the US Food and Drug Administration approved the first generation of cancer treatments delivered by nanoparticles (FDA).
Nanobiotechnology is gaining significant traction worldwide for cancer treatments and has been adopted to positively change laboratory procedures, thereby providing new ways for patient sample assessment and early detection of disease biomarkers with increased sensitivity and specificity.
One of the major advantages of nanoparticles lies in its ability to surface modification and encapsulation of poorly soluble anticancer drugs. Owing to its myriad applications in medical science, nanotechnology can positively shape the future of molecular diagnostics for cancer research.
At present, molecular diagnostics is rapidly advancing in both research and medicine, with the continuous integration of new technologies and applications for the rapid diagnosis of cancer. It has transformed cancer research by uncovering the molecular intricacies of the disease. It offers unprecedented opportunities for early detection, personalized medicine, and monitoring treatment response. As researchers continue to explore its potential, molecular diagnostics holds the key to advancing our understanding of cancer and improving patient care. With ongoing technological advancements, we can expect further breakthroughs that will revolutionize cancer research and pave the way for more effective treatments in the future.
Explore the power of molecular diagnostics in cancer research with Netscribes. Stay ahead of market trends and capitalize on business opportunities in the life science and healthcare realm. Contact us now to make well-informed decisions and shape the future of patient care.
Based on insights by Netscribes Life Sciences team